Effect of remineralization after in office followed by home treatment of white spot lesions in children randomized controlled trial
Study design
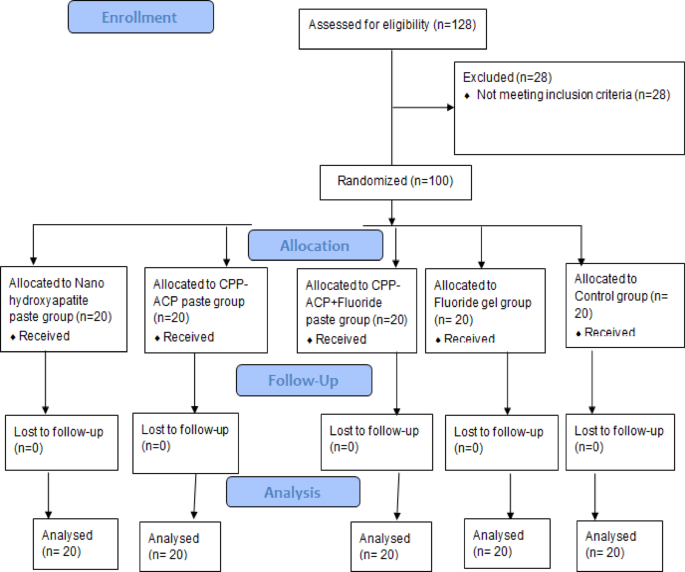
This study was conducted in accordance with the Consolidated Standards of Reporting Trials (CONSORT) 2025 statement for the planning and reporting of clinical trials19. A total of 40 participants were initially enrolled. Thirty-two patients, presenting with 100 anterior permanent teeth diagnosed with white spot lesions, were randomly assigned to different treatment groups. Participants then received the designated interventions and were evaluated for various outcomes at multiple time points. These procedures are detailed in the CONSORT 2025 flow diagram shown in (Fig. 1).

Participants’ teeth flow diagram.
Ethical approval and registration
This study had approval number 0342 on November 26, 2024 from the Medical Research Ethical Committee, National Research Centre, Egypt. The ethical standards of Helsinki stated in 1964 and its later amendments were strictly followed20. This study was initially registered on 12/02/2025 as first posted date, with ID: NCT06821724 -https://clinicaltrials.gov/study/NCT06821724.
Sample size calculation
Based on Ebrahimi et al. (2017)11, we determined that a total sample size of 100 teeth (20 per group) was adequate to detect a large effect size (f = 0.35), with an actual power (1-β error) of 0.8 (80%) and a significance level (α error) 0.05 (5%) for two-sided hypothesis test. using G*Power statistical analysis software (version 3.1.9.4)21.
Participants
Among children referred to the Department of Pedodontics, 40 children of both sexes, aged 10 to 14 years, were considered during the study period. Eligibility was assessed by the principal investigator, and eight children were excluded due to poor oral hygiene. Ultimately, 32 children were enrolled in the study according to the established inclusion criteria.
Inclusion criteria
Our study included “children between the ages of ten and fourteen with acceptable oral hygiene and brushed their teeth at least twice daily with no or limited number of cavitated teeth. Additionally, the children must have at least one anterior tooth affected by incipient carious lesion in the form of white spot lesion without any signs of cavitation by visual inspection and Diagnodent readings were between 5–17.”11,16.
Exclusion criteria
Children were excluded from the study if they were unwilling to be randomly assigned to any of the intervention groups; had abnormal physical, mental, or dental conditions; reported allergies to dairy products; presented with dentin caries, enamel hypoplasia, or restorations on the affected teeth; or had previously received treatment for WSLs22.
Setting and location
Children were selected from the Pedodontic clinics, National Research Centre, Egypt. Diagnosis was done in 2024 between November and December. The follow-up periods were fulfilled in February 2025.
Intervention
Informed consent
Just before the intervention, parents read well and signed the informed consent after summarization of the procedures in a simple way by the clinicians. Verbal approval was also gained from the child preoperatively.
Randomization
The participated children’s affected teeth in the study were randomly allocated into five groups (n = 20 each) using a 1:1 allocation ratio. Randomization was performed on 15 November 2024 via the website www.random.org. They were assigned to the five groups based on the remineralizing agent used, as follows:
-
Group I: Nano-hydroxyapatite (NHA) paste (Apagard Primo toothpaste).
-
Group II: Casein phosphopeptide-amorphous calcium phosphate (CPP-ACP) (MI Paste).
-
Group III: CPP-ACP with fluoride (MI Paste Plus).
-
Group IV: Fluoride gel (positive control group).
-
Group V: No treatment, only oral hygiene instructions provided (negative control group).
Each material was applied for five minutes within its respective group.
Allocation concealment
MF performed the random sequence generation using software available at The randomization table was securely maintained by MF to ensure complete allocation concealment. The operators, MM and RS, enrolled the participants, and MF revealed the group allocation for each child at the time of treatment. MM and RS administered the assigned interventions.
Blinding
In the current study, a double-blinded process was implemented, including assessors’ dentists, children, and biostatisticians. Treatment outcomes were evaluated by a professional expert who was blinded to the applied treatment for each subject.
Clinical steps
Diagnosis of white spot lesions
Visual examination
Clinically, a wet lesion appears as a white, opaque area due to optical changes resulting from mineral loss and the difference in refractive indices between air and water that fills the micro-pores formed in the WSL23.
Therefore, thorough enamel drying with air for at least five seconds was performed following tooth cleaning with pumice, to enable accurate examination under appropriate illumination24.
Laser fluorescence device “diagnodent
Demineralization assessment was conducted using a laser fluorescence device (DIAGNOdent®; KaVo). All measurements were performed using the type B tip, which is specifically designed for flat surfaces, such as the labial surfaces of anterior teeth.
In line with the manufacturer’s instructions, the device was calibrated via a standard ceramic plate before every recording16.
Following calibration, the tip of the DIAGNOdent device was kept in close contact with the tooth surface, while tilting it in the affected region to collect fluorescence from all angles. In this study, all teeth recorded values above 7 on the digital display, which indicates the presence of subsurface areas of demineralization on the tooth surface. When the DIAGNOdent score ranged between 3 and 7, this indicated a normal enamel surface25.
Two blinded external investigators recorded all measurements.
Grouping of samples
Group I: NHA paste was applied for five minutes.
Group II: CPP-ACP paste was applied for five minutes.
Group III: CPP-ACP with fluoride paste was applied for five minutes.
Group IV: Fluoride gel was applied for five minutes (positive control group).
Group V: No treatment was applied; only oral hygiene instructions were given (negative control group).
Treatment modalities
In-office application
Preoperative preparation
Prior to the initiation of any treatment, scaling and polishing were performed using a non-fluoridated paste. Isolation was achieved using cotton rolls and a saliva ejector. Lip and cheek retraction were performed using a cheek retractor.
Grouping of subjects
Group I: Nano hydroxyapatite paste.
The participants in this group were treated with Apagard Royal (Sangi Co., Ltd., Japan), containing 10% nanohydroxyapatite.
A thin layer of the paste was placed with a plastic applicator on the surface of the affected teeth for 5 min.
Group II: CPP-ACP paste.
Participants in this group were treated with a CPP-ACPF remineralising paste (MI Paste Plus; GC Corporation, Tokyo, Japan). The paste was applied following the same protocol described for Group I.
Group III: CPP-ACP + fluoride paste.
Participants in this group received a remineralizing paste containing CPP-ACPF and fluoride (MI Paste Plus; GC Corporation, Tokyo, Japan), which was applied in the same manner as described for Group I.
Group IV: Fluoride gel (positive control group).
The participants in this group were treated with a 1.23% acidulated phosphate fluoride gel (Dharma, IONITE, APF gels, USA).
A sufficient amount of gel was placed for 5 min on the affected area of the teeth. The gel was applied with a cotton swab to minimize its ingestion26.
Group V: no treatment (negative control).
No treatment was applied; only oral hygiene instructions were given (control group)27.
All children included in the study were instructed to brush their teeth using a toothbrush under the supervision of the operator prior to the application of the experimental materials.
Each agent was applied to the labial surface of each tooth affected by a WSL for five minutes, following cleaning and drying with a cotton roll. Application was performed using an applicator tip, followed by rinsing with water.
The children were instructed to stop eating or drinking for one hour after treatment.
Home application
Each participant received a sterilized pouch containing a sterile tube of the assigned remineralizing paste for home use. Parents were instructed to supervise the application and ensure that a pea-sized amount of paste was used twice daily until the end of the study.
Patients were instructed to perform the routine oral hygiene measures, and were given a pack with fluoridated toothpaste, a manual toothbrush, and dental floss. Cooperation and adherence to oral hygiene instructions were tested by questions at the recall visits concerning the rate of remineralizing agent application28.
These in-office procedures were repeated in the dental office, after one week, two weeks, and then four weeks.
Continuous and regular home application was followed from the first day of the study until its completion.
Outcome assessment
Laser fluorescence device “diagnodent
Remineralization assessment of white spot lesions (WSLs) was carried out using the DIAGNOdent laser fluorescence device immediately before application of the remineralizing agent and again ten minutes post-application. All measurements were performed in accordance with the manufacturer’s instructions and mirrored the method used during the initial diagnosis of WSLs.
Color assessment
Color change evaluation of WSLs was implemented via the Vita Easyshade spectrophotometer device (Vita Zahnfabrik, Bad Säckingen, Germany).
Prior to each measurement, calibration was done according to the manufacturer’s instructions. The tip was positioned at a right angle to the surface to ensure correct recording of tooth color. The measured readings were recorded according to the CIELAB (Commission International de l’Eclairage L*, a*, and b*) color space system. Based on this system, the “L” axis stands for the degree of lightness, whilst the “a” and “b” values indicate positions on the red/green (+ a = red, −a = green) and the yellow/blue (+ b = yellow, −b = blue) axes, respectively. Color assessment for all teeth was performed at the central area of WSL.
To minimize recording errors, each evaluation was repeated three times, and the mean value of three consecutive measurements was recorded for each area. Color assessments were repeated under identical conditions during the recall visits29.
The color change of WSLs from before to after the various remineralization treatments at each follow-up period was calculated using the following formula:
ΔE₀₀ (L*₁, a*₁, b*₁ ; L*₂, a*₂, b*₂) = ΔE₁₂₀₀ = ΔE₀₀.
Color evaluation of WSLs was recorded at baseline (before treatment), immediately after treatment, one week after, and one month after the start of treatment. This assessment was conducted on each tooth in both the study and control groups using the Vita Easyshade device.
Follow-up evaluations were conducted at one week and one-month post-treatment for each tooth in the corresponding study and control groups16.
Both remineralization and color assessments were recorded by two blinded external investigators.
Example of cases at different follow up periods are shown in (Figs. 2, 3, 4 and 5).

shows color change for a case in NHA paste group; (a) preoperatively, (b) Immediately after application, (c) after one month.

Shows color change for a case in MI paste group; (a) preoperatively, (b) Immediately after application, (c) after one month.

Shows color change for a case in MI Plus paste group; (a) preoperatively, (b) Immediately after application, (c) after one month.

Shows color change for a case in Fluoride group; (a) preoperatively, (b) Immediately after application, (c) after one month.
Statistical analysis
Numerical data were displayed as means with 95% confidence intervals (CI). They were analyzed for normality and variance homogeneity by inspecting the distribution and using Shapiro-Wilk and Levene’s tests, respectively. The data were noticed to be normally distributed; however, the homogeneity assumption was violated. They were analyzed using a heteroscedasticity-corrected (HC) two-way mixed model ANOVA, followed by simple effects comparisons of estimated marginal means while utilizing the error term of the main model. P-values were adjusted for multiple comparisons using the False Discovery Rate (FDR) method. Effect sizes were interpreted based on Cohen (1992)30. Correlation analysis was made using Spearman’s rank-order correlation coefficient. Statistical analysis was conducted with R statistical analysis software version 4.4.3 for Windows31.
link








